Command Palette
Search for a command to run...
外科 AI における比較研究:データセット、ファウンデーションモデル、および Med-AGI への障壁
外科 AI における比較研究:データセット、ファウンデーションモデル、および Med-AGI への障壁
概要
近年の人工知能(AI)モデルは、複数の生体医学タスクに関するベンチマークにおいて、人間の専門家の水準に匹敵、あるいはそれを上回る性能を示してきた。しかし、手術画像分析に関するベンチマークにおいては、依然として後れをとっている。手術は、マルチモーダルデータの統合、人間との相互作用、物理的効果の考慮など、多様なタスクの統合を必要とするため、その性能が向上すれば、汎用性の高い AI モデルは協働ツールとして特に有望であると考えられる。一方で、アーキテクチャの規模と訓練データの拡大という従来のアプローチは魅力的である。特に、年間数百万時間にわたる手術動画データが生成されている現状を踏まえると、その有効性は高まっている。他方、AI 訓練用に手術データを準備するには、専門的な知識が格段に必要とされ、そのデータを用いた訓練には莫大な計算資源を要する。これらのトレードオフは、現代の AI が手術実践をどの程度、あるいはどのように支援できるかについて、不確実な見通しをもたらしている。本研究では、2026 年時点で利用可能な最先端の AI 手法を用いた手術器具検出の事例研究を通じて、この問いを検証する。その結果、数十億パラメータを有する大規模モデルを用い、広範な訓練を実施しても、現在の Vision Language Models は、神経外科における器具検出という一見単純なタスクにおいてさえ、十分な性能を発揮できないことを示す。さらに、モデル規模と訓練時間の増大が関連する性能指標の向上に寄与する効果は逓減的であるというスケーリング実験の結果も明らかにした。これらの実験結果は、現状のモデルが手術用途において依然として重大な障壁に直面している可能性を示唆している。特に、一部の障壁は追加の計算資源による「スケーリング」では解決できず、多様なモデルアーキテクチャにわたって持続している。この事実は、データとラベルの可用性が唯一の制限要因であるのかという疑問を提起するものである。本稿では、これらの制約の主要な要因について議論し、潜在的な解決策を提示する。
One-sentence Summary
Researchers from Chicago Booth and the Surgical Data Science Collective demonstrate that scaling Vision Language Models fails to solve surgical tool detection, revealing that specialized architectures like YOLOv12-m significantly outperform billion-parameter systems in neurosurgery and laparoscopy despite massive computational investment.
Key Contributions
- The paper evaluates zero-shot surgical tool detection across 19 open-weight Vision Language Models on the SDSC-EEA neurosurgical dataset, revealing that despite increased model scale, only one model marginally exceeds the majority class baseline.
- A specialized classification head replacing off-the-shelf JSON generation is introduced for fine-tuned Gemma 3 27B, achieving 51.08% exact match accuracy and outperforming the baseline and standard fine-tuning approaches.
- Experiments demonstrate that a specialized 26M-parameter YOLOv12-m model achieves 54.73% exact match accuracy, outperforming all tested Vision Language Models while using 1,000 times fewer parameters and generalizing effectively to the CholecT50 laparoscopic dataset.
Introduction
Surgical AI aims to create collaborative tools capable of integrating multimodal data and physical effects to assist in complex procedures, yet current systems struggle to match human performance on surgical image-analysis benchmarks. While the prevailing scaling hypothesis suggests that increasing model size and training data will solve these issues, prior work faces significant challenges including the high cost of data annotation, the need for specialized expertise, and the risk that simply adding compute cannot overcome domain-specific distribution shifts. The authors leverage a case study on surgical tool detection to demonstrate that even multi-billion parameter Vision Language Models fail to surpass trivial baselines in zero-shot settings and show diminishing returns when scaled, ultimately proving that specialized, smaller models like YOLOv12-m outperform large foundation models with far fewer parameters.
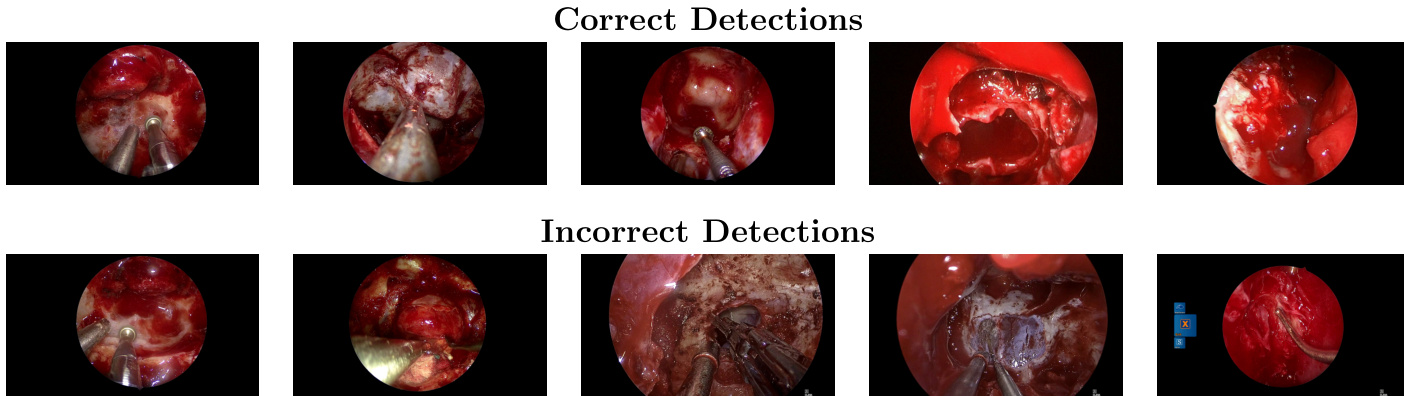
Dataset
-
Dataset Composition and Sources: The authors utilize the SDSC-EEA dataset, which contains 67,634 annotated frames extracted from 66 unique endoscopic endonasal approach (EEA) neurosurgical procedures. These video recordings were donated by 10 surgeons across 7 institutions in the United States, France, and Spain, with no exclusion criteria applied to the selection.
-
Key Details for Each Subset:
- Annotation Quality: Ground truth labels for 31 distinct surgical instrument classes were generated by three non-clinical annotators, reviewed by a senior annotator and SDSC members, with fewer than 10% of frames requiring correction.
- Format and Distribution: Annotations are provided in YOLO format with bounding boxes. The dataset shows significant class imbalance, with Suction appearing in 63.3% of frames, while other tools like Cotton Patty and Grasper appear less frequently.
- Split Strategy: To prevent data leakage, the data is split by surgical procedure rather than individual frames. This results in a training set of 47,618 frames from 53 procedures and a validation set of 20,016 frames from 13 procedures.
-
Usage in the Model:
- Fine-tuning: The training split is used for LoRA fine-tuning of the Vision-Language Model (VLM).
- Zero-Shot Evaluation: The authors evaluate zero-shot VLM performance using a specific prompt template that lists all 31 valid tool names and requires the model to return detected tools in a strict JSON format.
- External Validation: The methodology includes validation on the external CholecT50 dataset to assess generalizability.
-
Processing and Metadata Details:
- Data Leakage Prevention: The procedure-level split ensures that frames from the same surgery never appear in both training and validation sets, leading to uneven tool distributions across splits (e.g., the Sonopet pineapple tip appears only in the training set).
- Labeling Protocol: Annotators received tool descriptions and representative images prior to labeling to ensure consistency, and the final dataset includes multi-label ground truth indicating the presence or absence of instruments in each frame.
Experiment
- Zero-shot evaluation of 19 open-weight vision-language models across two years of development shows that even the largest models fail to surpass a trivial majority class baseline for surgical tool detection, indicating that general multimodal benchmark performance does not transfer to specialized surgical perception.
- Fine-tuning with LoRA adapters improves performance over zero-shot baselines, with a dedicated classification head outperforming autoregressive JSON generation, yet a persistent gap between training and validation accuracy reveals limited generalization to held-out procedures.
- Scaling LoRA adapter rank by nearly three orders of magnitude saturates training accuracy near 99% while validation accuracy remains below 40%, demonstrating that the performance bottleneck is caused by distribution shift rather than insufficient model capacity.
- A specialized 26M-parameter object detection model (YOLOv12-m) outperforms all fine-tuned vision-language models on the primary dataset while using over 1,000 times fewer parameters, suggesting that task-specific data and architecture are more critical than model scale.
- Replication on an independent laparoscopic dataset (CholecT50) confirms that zero-shot performance remains poor, fine-tuning is necessary for high accuracy, and smaller specialized models continue to outperform large foundation models, including proprietary frontier systems.
- The overall findings suggest that progress in surgical AI is currently constrained by the availability of large-scale, standardized domain-specific data rather than the scale of AI architectures, pointing toward hybrid systems that combine generalist models with specialized perception modules.