Command Palette
Search for a command to run...
Fighting Chemotherapy Resistance and Tumor Recurrence! Shandong University Research Team Uses AI to Build a Powerful Defense Line for Breast Cancer Stem Cells

Breast cancer is the number one killer among female malignant tumors and has always been the focus of the medical community. Currently, cytotoxic chemotherapy is the most common systemic treatment for breast cancer patients. Although it can alleviate the condition to a certain extent, cancer cells may still recur or metastasize.
Previous studies have shown thatBreast cancer stem cells (BCSCs) are the source of breast cancer resistance to chemotherapy and radiotherapy. Although BCSCs only account for a small proportion of breast cancer cells, they have an amazing unlimited proliferation potential and the ability to initiate tumors. Even more shocking is that the chemotherapy process itself may also prompt non-BCSC cells to transform into BCSCs, thereby increasing the risk of tumor recurrence and metastasis.
It can be seen that a deep understanding of the response mechanism of BCSC to chemotherapy is very important for improving the prognosis of breast cancer patients. Clinically, a method that can identify and inhibit BCSC is urgently needed to optimize existing treatment options.
Faced with this challenge,Lv Haiquan, Sun Rong, Zhang Kai from Shandong University and Mei Qi from Shanxi Medical University, together with research teams from Helix Matrix and others, have made a breakthrough. Using machine learning technology and based on mRNA analysis, they have successfully developed a new method, BCSC signature, to assess the characteristics of cancer stem cells in samples from primary breast cancer patients.This study not only reveals the core role of polyamine anabolism in BCSC regulation, but also provides new strategies and directions for the clinical treatment of breast cancer.
The study was published in the internationally renowned journal Advanced Science under the title "Polyamine Anabolism Promotes Chemotherapy-Induced Breast Cancer Stem Cell Enrichment".
Research highlights:
* Researchers developed an mRNA-based BCSC signature using machine learning methods to assess cancer stemness in breast cancer patient samples
* This study found that polyamine anabolism plays a key role in BCSC regulation. Chemotherapy promotes the enrichment of BCSC by activating the polyamine anabolism pathway regulated by HIF-1.
* This study discovered a new specific HIF-1 inhibitor, Britannin, which can effectively inhibit chemotherapy-induced HIF-1 transcriptional activity, polyamine metabolism levels, and BCSC enrichment when used in combination

Paper address:
https://onlinelibrary.wiley.com/doi/10.1002/advs.202404853
Using TCGA datasets to build associations between Pearson correlation analysis and machine learning algorithms
To quantitatively evaluate the stem cell properties of primary breast cancer patient samples by gene expression levels.The study used data from The Cancer Genome Atlas Consortium (TCGA) on invasive breast cancer. The BRCA1 (BRCA2) dataset was used to correlate mRNA expression with the mRNA stem cell index (mRNAsi) based on the univariate logistic regression (OCLR) machine learning algorithm using Pearson correlation coefficient analysis, thereby developing an mRNA-based BCSC signature.
The study also generated Kaplan-Meier curves based on a data set of 2,032 breast cancer patients, partly stratified by the expression of BCSC P-Sig and N-Sig in primary tumors, and partly based on a data set of 1,372 breast cancer patients who received chemotherapy.
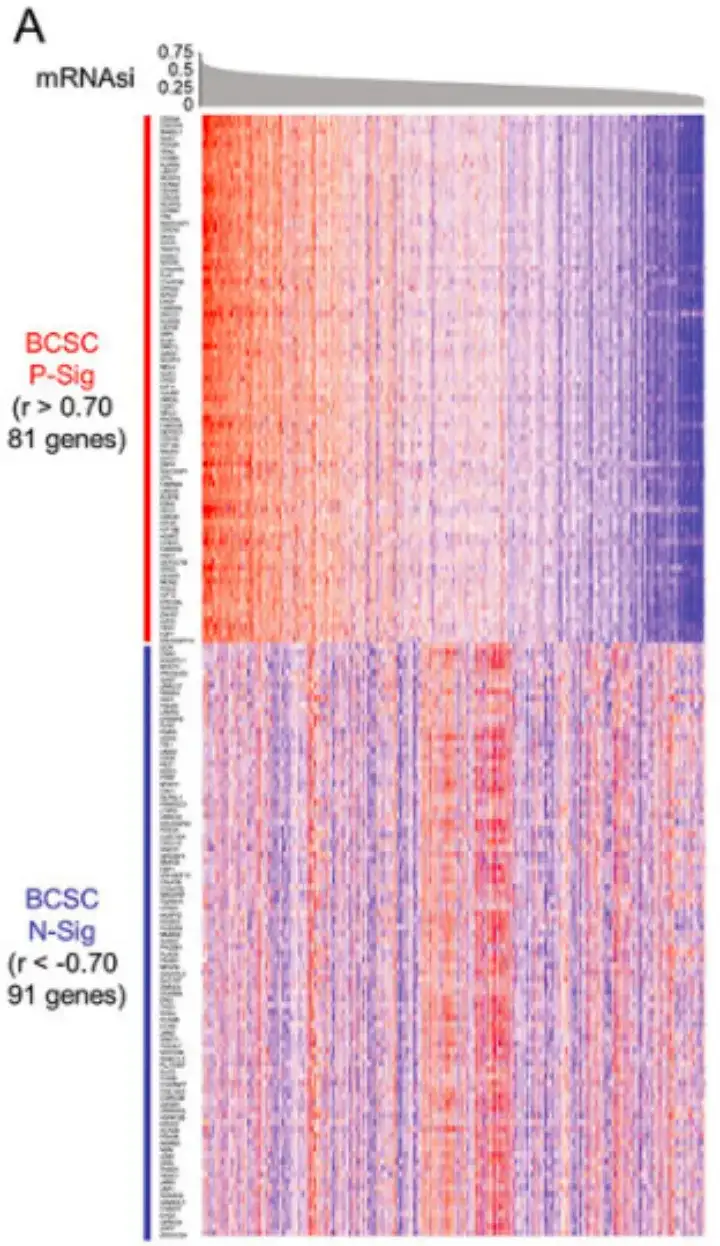
Specifically, as shown in Figure A below, this study first identified 81 genes with Pearson correlation coefficients r>0.70 and 91 genes with Pearson correlation coefficients r<-0.70, and defined them as BCSC positive signature (P-Sig) and negative signature (N-Sig), respectively.
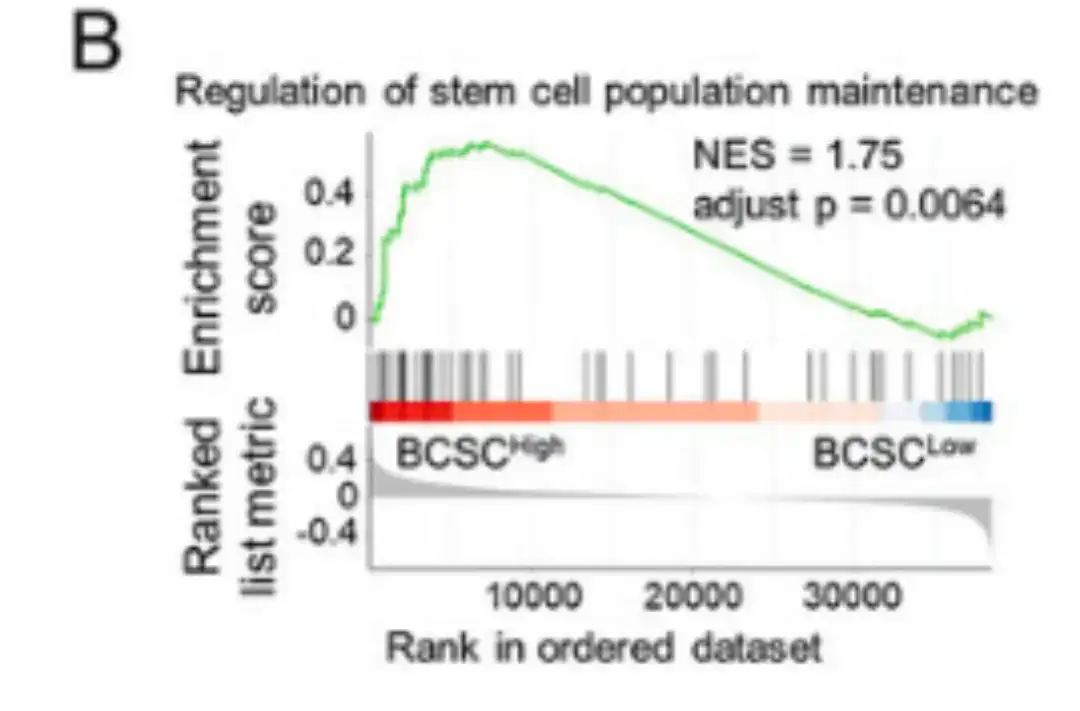
Subsequently, as shown in Figure B below, the study performed a ranking-based gene set enrichment analysis (GSEA) by comparing the BCSChigh and BCSClow data sets and stratifying the groups based on the expression of the BCSC signature.
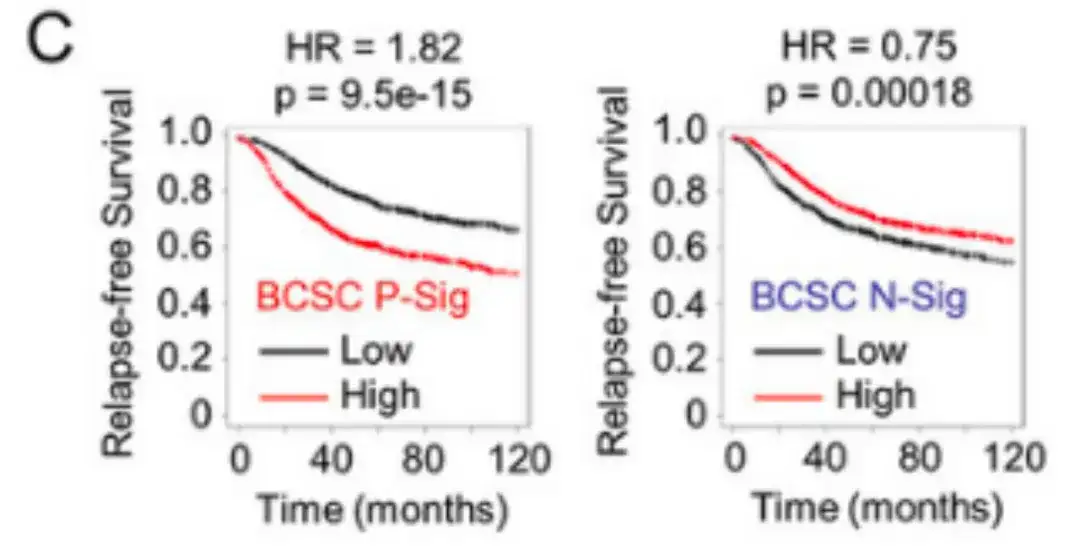
After analyzing the correlation between BCSC signature expression and recurrence-free survival (RFS) in 2,032 patients, the researchers found that patients with BCSC P-Sig expression above the median had significantly reduced RFS, while patients with BCSC N-Sig expression above the median had increased RFS, as shown in Figure C below.
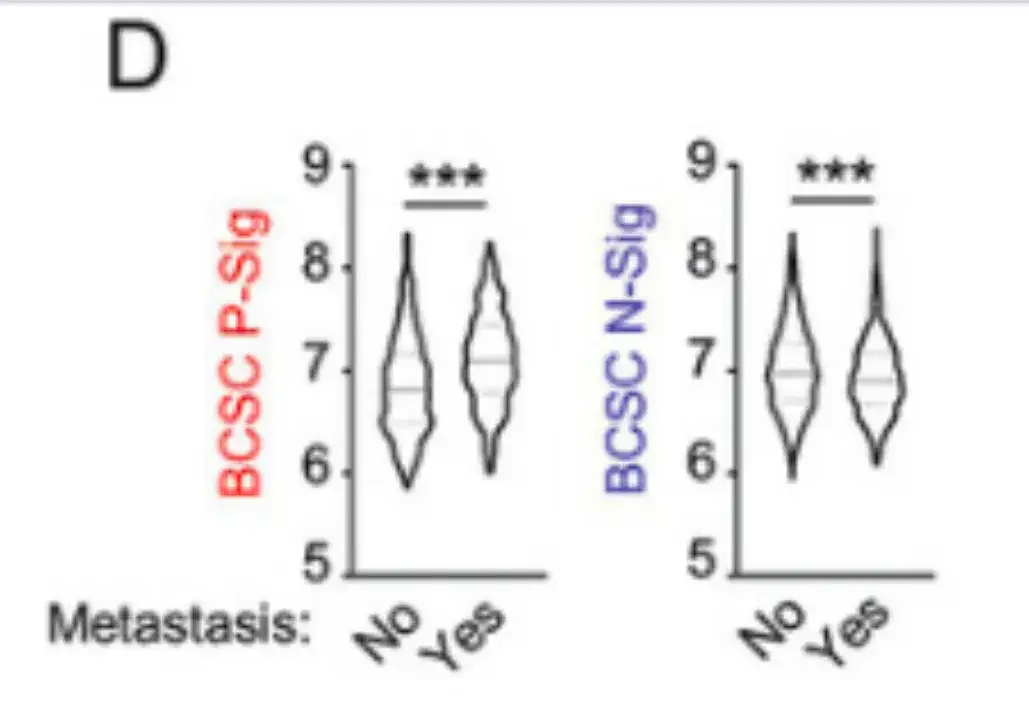
The study also analyzed data from 1,979 breast cancer patients in the International Breast Cancer Consortium's Molecular Classification Database (METABRIC).The results are shown in Figure D below. Patients who developed metastasis within 5 years had higher BCSC P-Sig expression and lower BCSC N-Sig expression in their primary tumors compared with patients who did not develop metastasis.
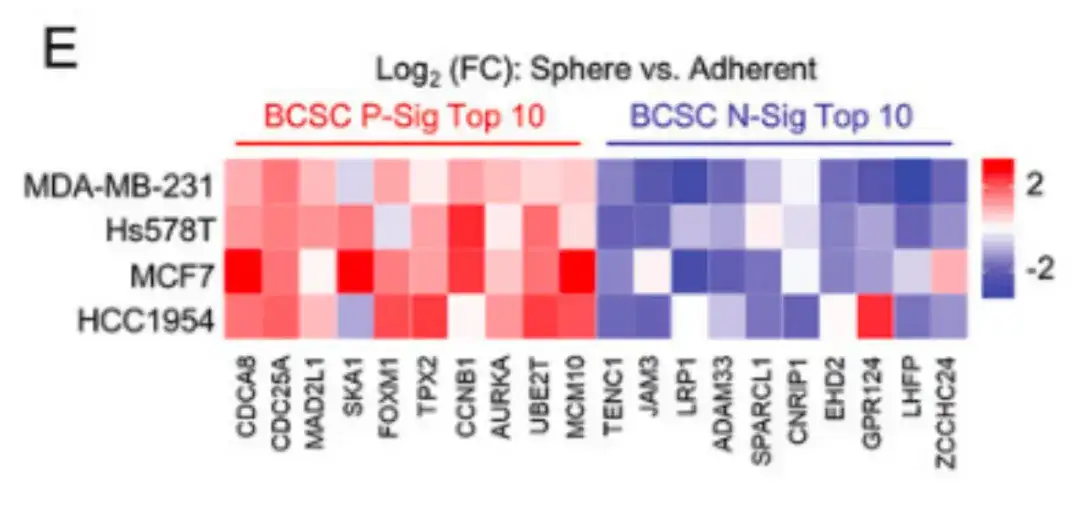
In addition, in Figure E below,The study also verified the BCSC characteristics in multiple breast cancer cell lines.Breast cancer cells cultured in mammosphere culture medium showed higher expression of BCSC P-Sig and lower expression of BCSC N-Sig compared to cells cultured in standard polystyrene tissue culture dishes.
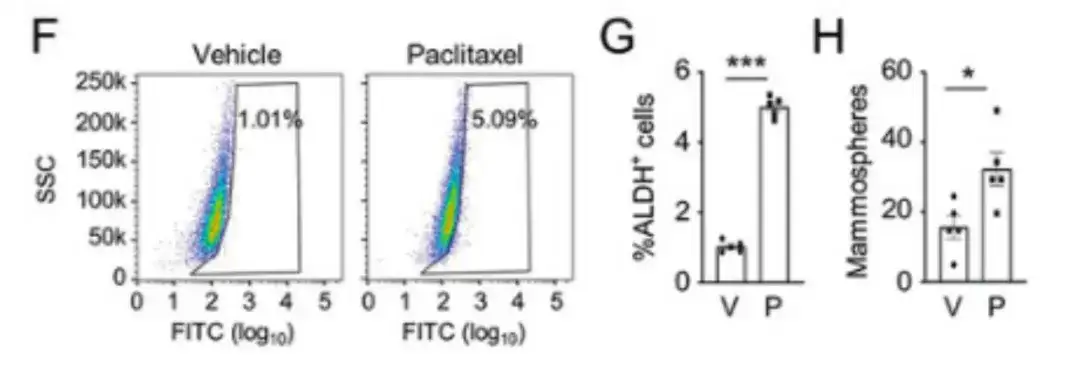
To evaluate how chemotherapy drugs affect the BCSC population,In this study, human breast cancer cells, MDA-MB-231, were injected into the mammary fat pad (MFP) of female severe combined immunodeficient (SCID) mice and the mice were treated with paclitaxel. As shown in Figures F, G, and H below, paclitaxel significantly increased the percentage of aldehyde dehydrogenase active (ALDH+) cells and the number of mammosphere-forming cells.
Meanwhile, in Figure I below, RNA-seq analysis showed that paclitaxel treatment increased the expression of most BCSC P-Sig genes and decreased the expression of most BCSC N-Sig genes.
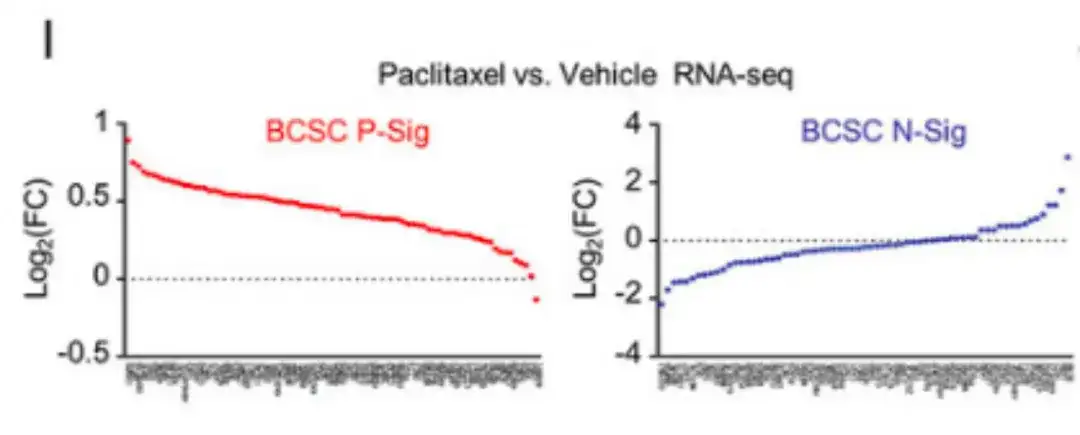
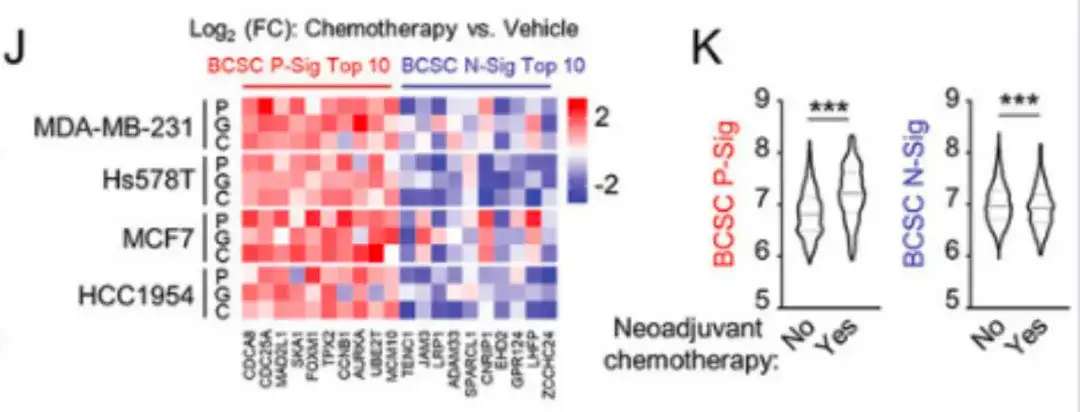
In addition to paclitaxel, in Figure J below, the study also treated breast cancer cells with FDA-approved chemotherapy drugs gemcitabine and carboplatin.These drugs increased the expression of BCSC P-Sig gene and decreased the expression of BCSC N-Sig gene.As shown in Figure K below, samples from METABRIC patients also showed that in patients receiving neoadjuvant chemotherapy, the expression of BCSC P-Sig gene in primary tumor tissue was higher, while the expression of BCSC N-Sig gene was lower.
Polyamine anabolism is positively correlated with BCSC enrichment, and the HIF-1 inhibitor Britannin can reduce polyamine biosynthesis and eradicate BCSC
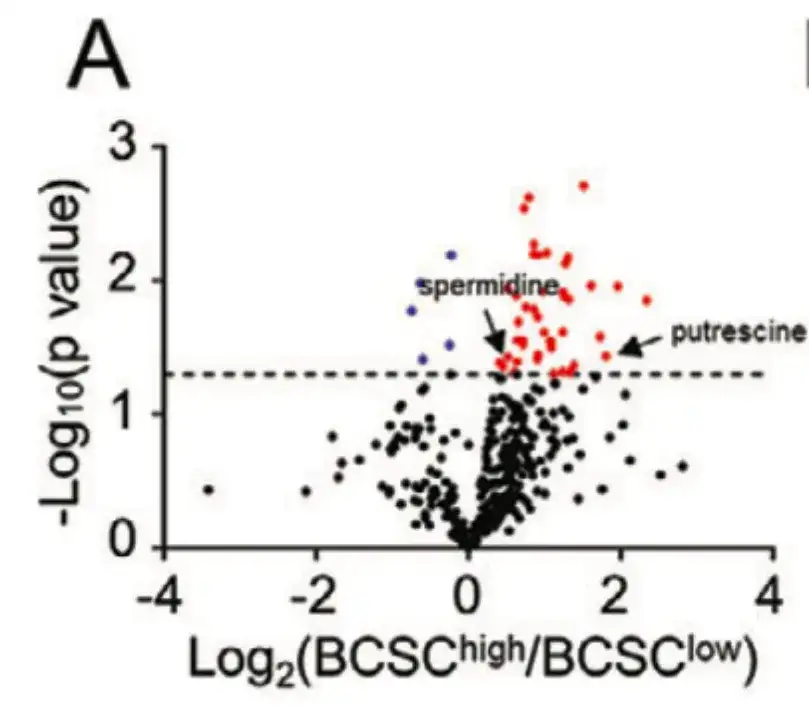
In order to explore the mechanism of chemotherapy-induced BCSC enrichment from the perspective of cell metabolism,This study divided 21 patients in TCGA BRCA into BCSChigh and BCSClow groups based on the expression of BCSC signatures, and compared the levels of 399 metabolites between the two groups, as shown in Figure A below.
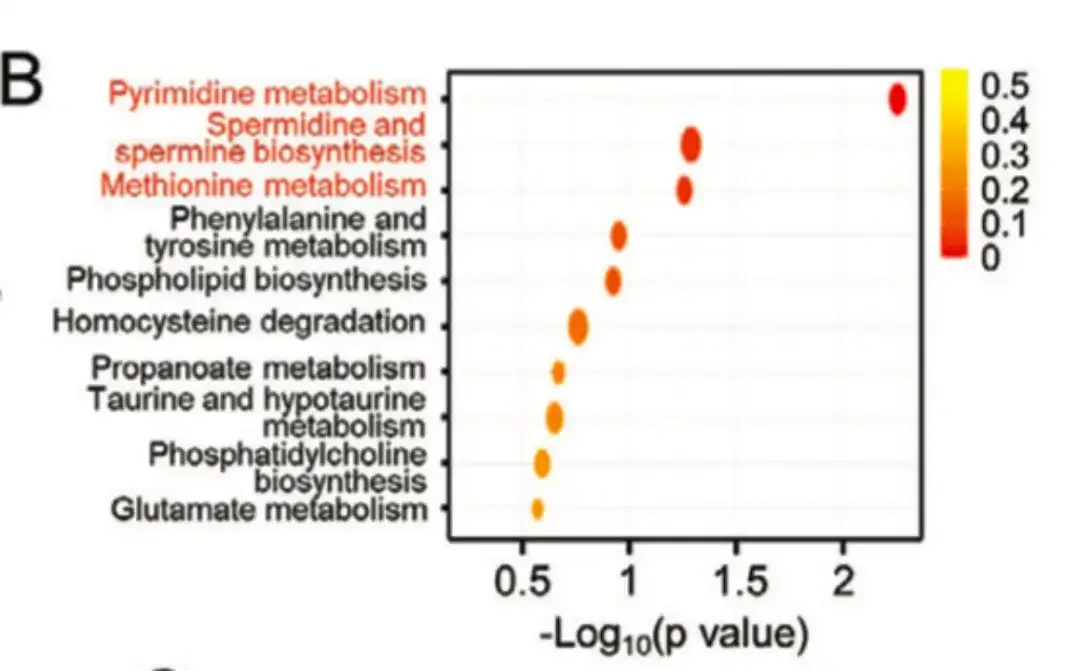
As shown in Figure B below, this study demonstrated through metabolic pathway enrichment analysis that metabolites with different levels of enrichment were formed in polyamine anabolic metabolism-related pathways.
In the following figure C, D,The study found that key enzymes of the polyamine biosynthesis pathway, such as ornithine decarboxylase 1 (ODC1), human spermidine synthase (SRM), and spermine synthase (SMS), were more highly expressed in the BCSChigh group than in the BCSClow group.And the expression in spheroid cultures grown on non-adherent breast cancer cell lines was higher than that in monolayer cultures, indicating that polyamine biosynthesis plays an important role in the regulation of BCSC phenotype. Meanwhile, as shown in Figure E below, paclitaxel treatment increased the expression of ODC1 and SRM in breast cancer cell lines.
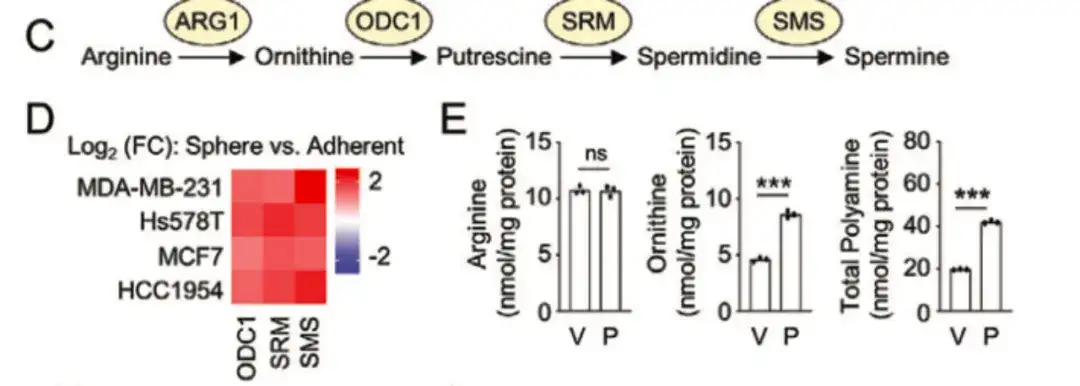
In addition, in Figure G below,The study also collected 34 desensitized breast cancer patient samples from Qilu Hospital of Shandong University.Analysis of the data revealed that the levels of ornithine and polyamines in cells of patients receiving neoadjuvant chemotherapy were significantly increased. These studies all indicate that enhanced polyamine anabolism promotes chemotherapy-induced BCSC enrichment.
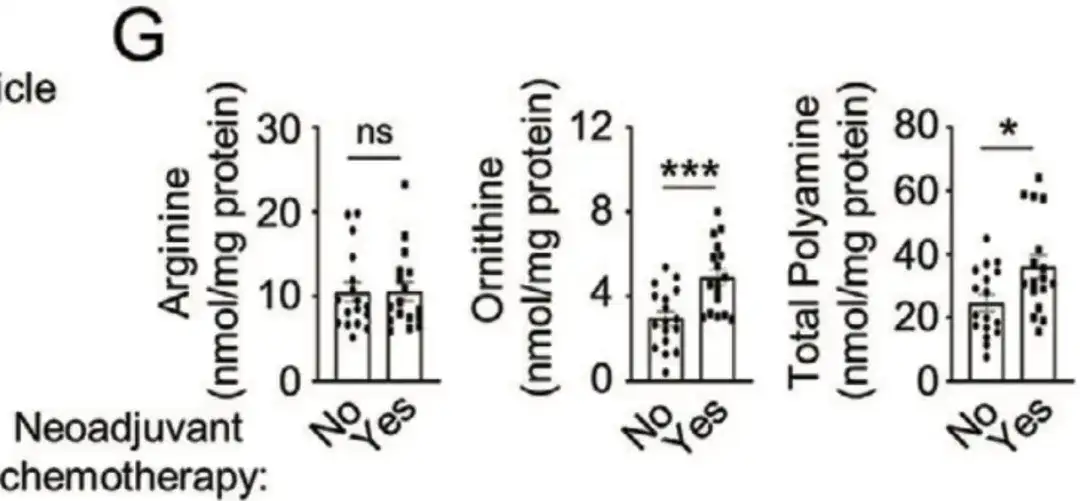
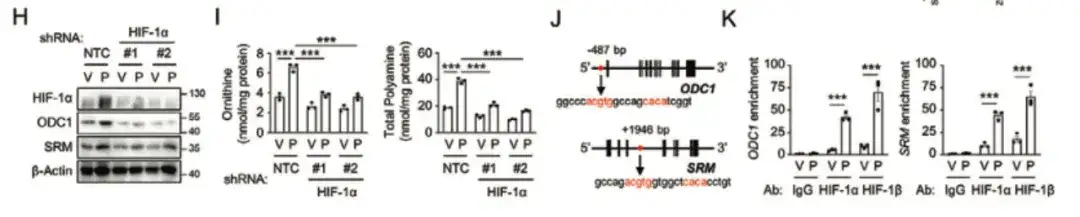
In order to further analyze the molecular mechanism by which chemotherapy promotes polyamine anabolism in breast cancer, the study found thatInhibition of hypoxia-inducible factor (HIF-1α) attenuated paclitaxel-mediated increase in ALDH+ and spheroid formation.It decreased the expression of most BCSC P-Sig genes and increased the expression of most BCSC N-Sig genes, indicating that HIF-1 plays a key role in regulating paclitaxel-induced BCSC enrichment, as shown in Figures AC below.
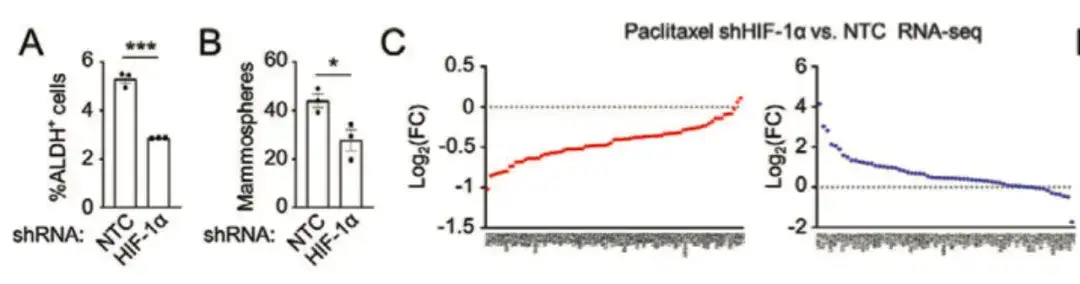
To further investigate the role of HIF-1 in regulating BCSC phenotypes, as shown in Figure DG below, the study also transfected MDA-MB-231 with the HIF-1 expression fluorescence tracking system. By collecting tumor tissues from mice treated with paclitaxel, the cells were divided into DsRed+/GFP− (HIF-1−) and GFP+ (HIF-1+) cell populations.Compared with the DsRed+/GFP− cell population, the GFP+ cell population with high HIF-1 expression and transcriptional activity had increased BCSC P-Sig gene expression and decreased BCSC N-Sig expression. Meanwhile, the tumorigenic ability of GFP+ cells was significantly higher than that of the untreated group or DsRed+/GFP− cells.
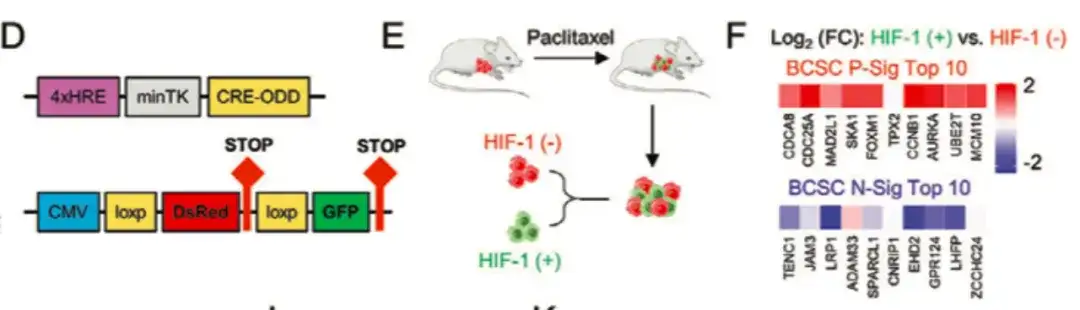
In Figures H and I below, the study found that inhibition of HIF-1α blocked paclitaxel-induced ODC1 and SRM expression, indicating that paclitaxel promoted polyamine anabolism in a HIF-1-dependent manner.
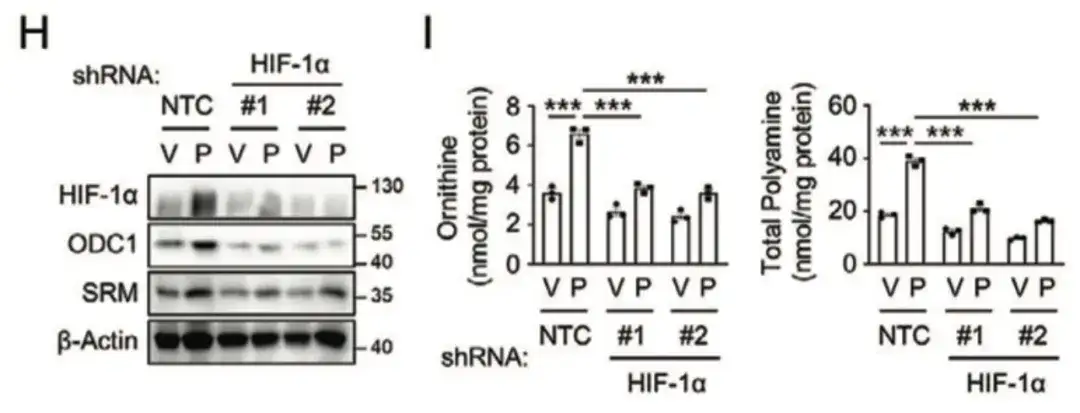
Finally, the study found that HIF-1 activated the ODC1 and SRM genes through transcription, promoting the anabolism of polyamines, which in turn led to the enrichment of chemotherapy-induced BCSCs, as shown in Figure LQ below.

As shown in Figure A below, the study used the HIF-1 transcriptional activity reporter system to transduce SUM159 cells and screened for new HIF-1 inhibitors in a natural product library containing 4,320 natural compounds. The study found thatBritannin from Inula aucheriana effectively inhibited the transcriptional activity of HIF-1 in a dose-dependent manner. The combined use of Britannin and paclitaxel could dose-dependently block the increase in intracellular arginine and polyamine levels caused by paclitaxel.As shown in Figure BF below.
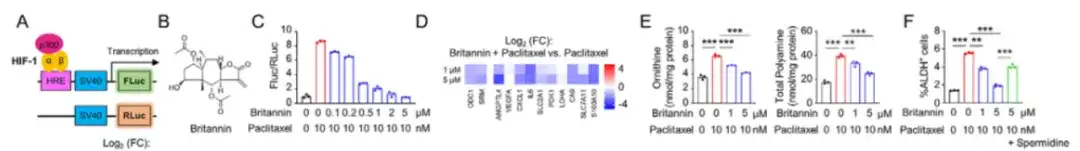
To investigate the mechanism by which Britannin inhibits HIF-1 transcriptional activity, in Figure HK below, the study co-transfected MDA-MB-231 cells with the HIF-1 transcriptional activity reporter system.The results showed that Britannin could attenuate the paclitaxel-induced HIF-1α TAD activity reporter system in a dose-dependent manner, and Britannin could inhibit the transcriptional activity of HIF-1 by blocking the transcriptional activator p300.
In the figure below, LQ, the study injected 2×106 MDA-MB-231 cells into SCID mice and treated the mice with paclitaxel alone or in combination with Britannin. The results showed that the combined treatment could significantly inhibit the growth of primary tumors without affecting the appearance and weight of the mice, and 3 out of 10 mice had no tumor recurrence 100 days after stopping drug treatment.This suggests that combined treatment of paclitaxel and Britannin in mice can completely eradicate BCSCs.

AI helps pathological diagnosis, and HER2-targeted therapy may be the key
Nowadays, the diagnosis of breast cancer not only relies on imaging results, but pathological diagnosis also plays an irreplaceable role. The expression status of HER2 (human epidermal growth factor receptor 2) is an important consideration in the treatment of breast cancer. AI can assist in identifying the expression level of HER2 in the analysis of pathological sections and provide a reference for subsequent targeted therapy.
Although the principle seems simple, it is not easy to achieve. For example, in order to use AI technology to improve the accuracy and repeatability of HER2 interpretation in breast cancer, a lot of training is required, and the AI system needs to be constantly debugged and compared with the diagnosis of medical personnel. On this basis, the researchers focused on the distinction between HER2 negative and positive, focusing AI on the low expression limit of HER2, so as to evaluate the value of AI in the diagnosis of different heterogeneous HER2 low-expression breast cancers.
In fact,As early as 2022, Professor Lv Haiquan of Shandong University published a cover article in the biomedical journal Theranostics, revealing that targeting A2BR combined with chemotherapy may block the enrichment of breast cancer stem cells and improve the survival rate of breast cancer patients after chemotherapy.
Paper link:
https://www.thno.org/v12p2598.htm

This time, Professor Lv Haiquan once again revealed that the combined use of Britannin can inhibit the polyamine anabolism of HIF-1 and eradicate BCSC, which undoubtedly provides a new idea for the treatment of breast cancer.In the future, AI will play an unprecedented and important role in the treatment of breast cancer, and its application in the medical and health field is increasingly showing great potential. With the continuous advancement of technology and in-depth application, AI will become an indispensable partner in breast cancer and even cancer treatment, bringing more strength and hope to patients.