Command Palette
Search for a command to run...
Can Inhibit Cancer Cell Proliferation! Huihu School of Pharmacy and Tianjin Medical University Jointly Developed a New Tumor Suppressor Protein Degrader dp53m

Maybe many people don’t know,In fact, each of us has cancer cells in our body.The human body undergoes the creation and replacement of billions or even tens of billions of cells every day. In this metabolic process, DNA replication is bound to go wrong, such as gene mutations that turn normal cells into cancer cells in situ.There are some tumor suppressor proteins in the human body.Such as p53, these proteins can curb the occurrence of cancer by regulating the cell cycle and inducing apoptosis or senescence of cancer cells. This is also an important reason why most of us can "coexist peacefully" with cancer cells.
The tumor suppressor protein p53, known as the "guardian of the genome", is encoded by the gene TP53 and plays an important role in preventing the occurrence of cancer.However, TP53 can undergo missense mutations at several common specific hotspots such as R175, G245, R248, R273 and R282, which in turn produce p53 mutants, leading to the loss of normal tumor suppressor protein function. In addition, due to the dominant negative effect of some p53 mutants, not only will the mutant p53 lose its original tumor suppressor function, but it will also interfere with the tumor suppressor activity of normal wild-type p53 (p53-WT), thereby increasing the risk of tumorigenesis.
Compared with several other p53 mutants,The mutant p53-R175H protein has a higher potential for tumorigenesis, metastasis, and drug resistance.Developing drugs targeting p53-R175H and allowing targeted drugs to accurately recognize and degrade p53-R175H is an effective strategy to inhibit cancer. However, since most mutant p53 proteins lack active sites for small molecule drugs, it is extremely difficult to design targeted drugs that can accurately recognize and affect mutant p53 proteins.
In this regard,Professor Wu Sijin from the Huihu School of Pharmacy at Xi'an Jiaotong-Liverpool University, together with Professor Xie Songbo and Professor Zhong Diansheng from Tianjin Medical University General Hospital,A paper titled “An engineered DNA aptamer-based PROTAC for precise therapy of p53-R175H hotspot mutant-driven cancer” was published in Elsevier.
This study developed a selective p53-R175H degrader, dp53m.This degrader can specifically recognize the mutant p53-R175H protein and utilize the natural protein degradation system in the cell - the ubiquitin protease system to achieve targeted degradation of the target protein and inhibit the functional expression of the mutant p53 protein.
The degrader has significant anti-tumor efficacy and no obvious toxicity. In addition, dp53m can also work synergistically with the chemotherapy drug cisplatin to enhance the sensitivity of cancer cells to cisplatin, which is crucial for the treatment of cancer.
Research highlights:
- The researchers modified non-core bases under the guidance of MD simulations and guided the RNA aptamer to a DNA aptamer (p53m-DA) by iterative molecular docking-guided post-SELEX method.
- As a component of dp53m, p53m-DA can specifically recognize p53-R175H protein. CRBN is involved in protein degradation, so dp53m can specifically recognize p53-R175H protein and degrade it.

Paper address:
https://pubmed.ncbi.nlm.nih.gov/38811338
The open source project "awesome-ai4s" brings together more than 100 AI4S paper interpretations and also provides massive data sets and tools:
https://github.com/hyperai/awesome-ai4s
Post-SELEX engineering: Construction of a high-performance DNA aptamer that specifically recognizes mutant p53-R175H
In the study of p53, researchers have found that the R175H mutation affects the DNA binding function by changing the structural state of p53. The combination of multiple inhibitor small molecules and aptamers can partially restore the activity of p53-R175H, but the molecular mechanism of its influence has not been reported.
This study first used molecular simulation methods to explore the intrinsic molecular mechanism of aptamers in restoring the function of p53-R175H.Compared with normal p53-WT, in mutant p53-R175H, the distance between L3 and C-helix is closer, and the distance between L2 and L3 is larger. The structural changes at these positions may be the main reason why the p53-R175H mutant loses its typical function, and also provides a structural basis for the specific recognition of its aptamers and related PROTAC molecules.
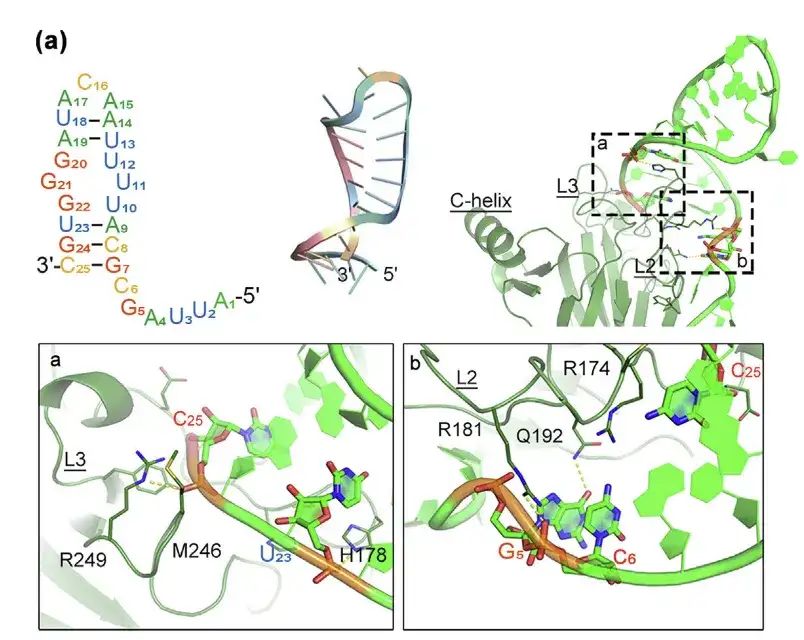
- Prediction of the structure of p53m-RA and its interaction with p53-R175H protein using molecular modeling
- The bonding interface is shown in regions a and b.
In previous studies, the RNA aptamer targeting p53-R175H (p53m-RA) has been shown to have high affinity for p53-R175H, however, it is very unstable in serum, which limits its practical application.
References:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9884801
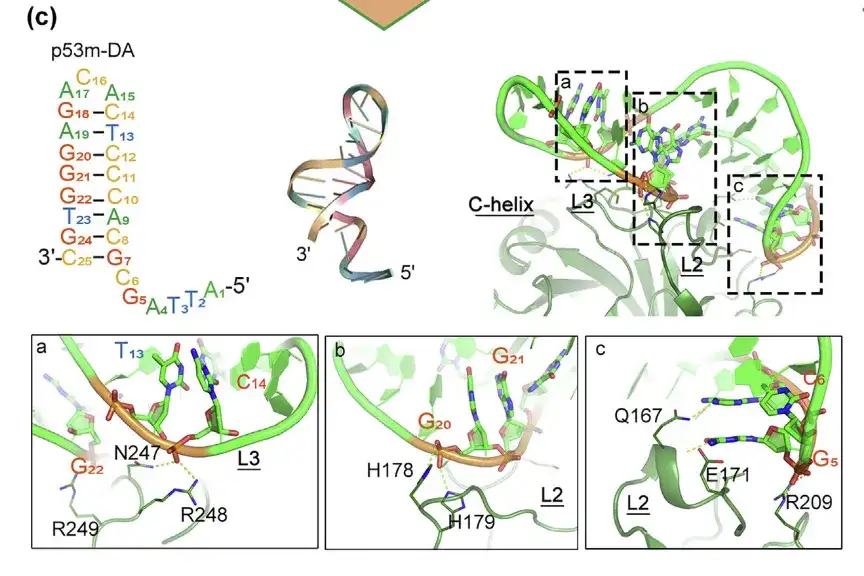
Post-SELEX optimized p53m-DA and its interaction with p53-R175H
The bonding interface is in regions a, b, and c
To enhance the structural stability and affinity of the aptamer, the researchers modified the non-core bases under the guidance of MD simulation and engineered the RNA aptamer into a DNA aptamer (p53m-DA) through post-SELEX.
The computational power of the molecular simulations in this study was provided by OpenBayes supply
The results showed that the structure of the paired region of p53m-DA is a major groove, and the unpaired loop region is a minor groove. The major groove plays a key role in binding with H178, H179, E171 and Q167 in L2, and the minor groove plays a key role in interacting with N247, R248 and R249 in L3.In addition, p53m-DA continuously maintained a double-stranded helical conformation throughout the simulation, which indicates the stability of its structure.
L2 and L3 are regions present in the mutant protein
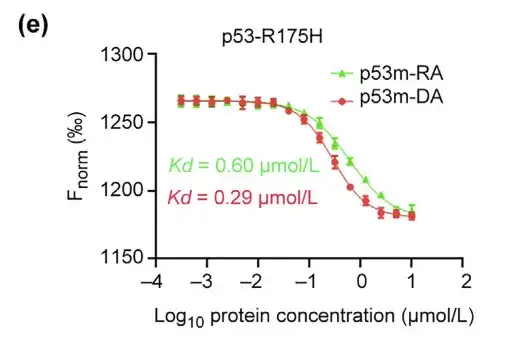
The researchers further evaluated the affinity and selectivity of p53m-DA for p53-R175H. MST assay showed that the Kd of p53m-DA was 0.29 lmol/L, which was about 2 times lower than that of p53m-RA.
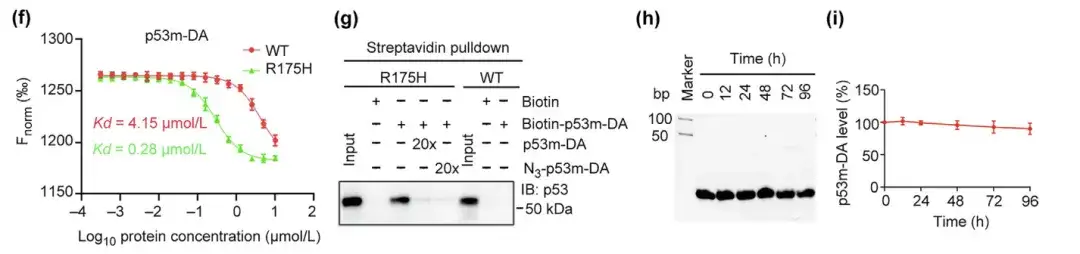
(f) Affinity of p53m-DA for p53-WT and p53-R175H
(g) Specific binding of p53m-DA to p53-R175H by streptavidin pulldown assay
(h,i) Evaluation of serum stability of p53m-DA and determination of the residual amount of p53m-DA
In addition, p53m-DA showed a 14-fold higher affinity for mutant p53-R175H than for normal p53-WT. In the streptavidin pulldown assay, p53m-DA specifically bound to p53-R175H and did not interact with p53-WT, demonstrating the specificity of p53m-DA for p53-R175H. Notably, the serum stability of p53m-DA was significantly improved, with minimal degradation after 96 hours.
In summary,p53m-DA is an aptamer with high specificity and stability.
dp53m: a specific p53-R175H degrader
Experiment 1: Synthesis of DP53M
The 5' end of the aptamer p53m-DA is located outside its binding site with p53-R175H. The researchers used a click reaction to conjugate alkynyl thalidomide (a ligand of CRBN E3 ligase) to the 5' end of N3-p53m-DA. The resulting protein degradation targeting chimera PROTAC (named dp53m) can selectively bind to p53-R175 H. The structure of dp53m is shown in the figure below:
*PROTAC: consists of an E3 ligase ligand, a ligand targeting POI, and a chemical linker between the ligands
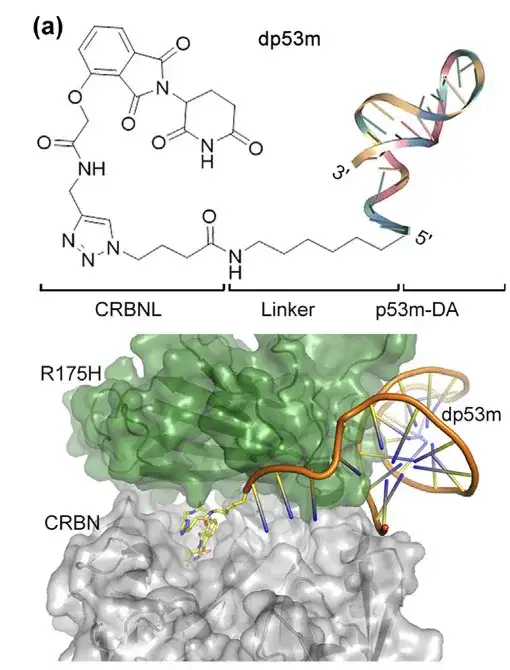
(a) The structure of dp53m was simulated using protein-protein docking and molecular dynamics calculations.
Bottom: Its binding interface with p53-R175H and CRBN
Experiment 2: dp53m can specifically recognize and degrade mutant p53-R175H
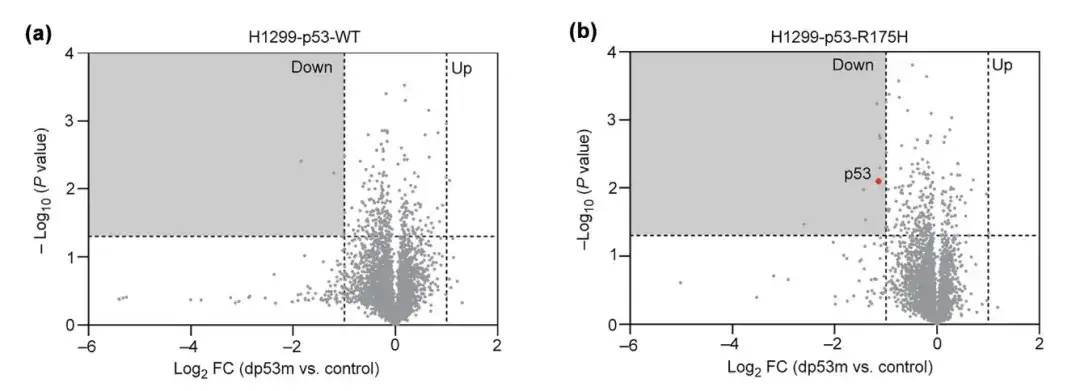
To study the cell specificity of dp53m for p53-R175H, the researchers treated p53-R175H and p53-WT overexpressed in H1299 cells with PBS or dp53m for 16 hours and monitored their protein changes. The results showed that dp53m significantly degraded p53-R175H, but not p53-WT.
*PBS is phosphate buffered saline, used as control group
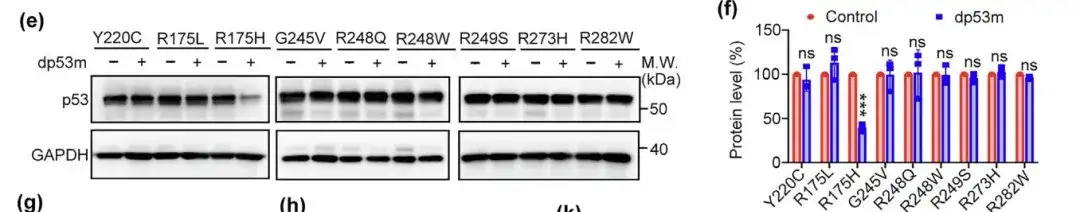
In addition, the researchers also evaluated the degradation potential of dp53m on other hotspot mutants such as Y220C, R175L, G245V, R248Q, R248W, R249S, R273H and R282W. The results showed that dp53m only effectively degraded p53-R175H,Its specificity for degradation of p53-R175H was demonstrated.
Compared with p53WT, dp53m treatment increased the polyubiquitination of p53-R175H. Studies have shown that the degradation of dp53m can be blocked by the protease inhibitor MG132, CRBN ligand or CRBN-targeting siRNA.It is speculated that dp53m-induced degradation of p53-R175H occurs through the ubiquitin-proteasome mechanism.
In summary,dp53m is a specific p53-R175H degrader, and its degradation process occurs through the ubiquitin-proteasome mechanism.
dp53m inhibits cancer cell proliferation and migration and tumor growth
Experiment 1: dp53m inhibits cancer cell proliferation and migration induced by p53-R175H
The researchers treated H1299-p53-WT, H1299-p53-R175H, A549, Detroit 562 and SKBR3 cells with PBS, p53m-DA or dp53 m, respectively, performed viability assays on them, and observed colony formation in the cells.
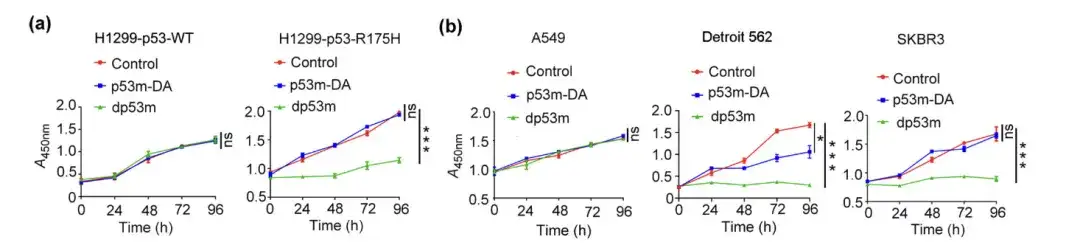
Viability assay of H1299-p53-WT, H1299-p53-R175H, A549, Detroit 562, and SKBR3 cells treated with PBS, p53m-DA, or dp53m
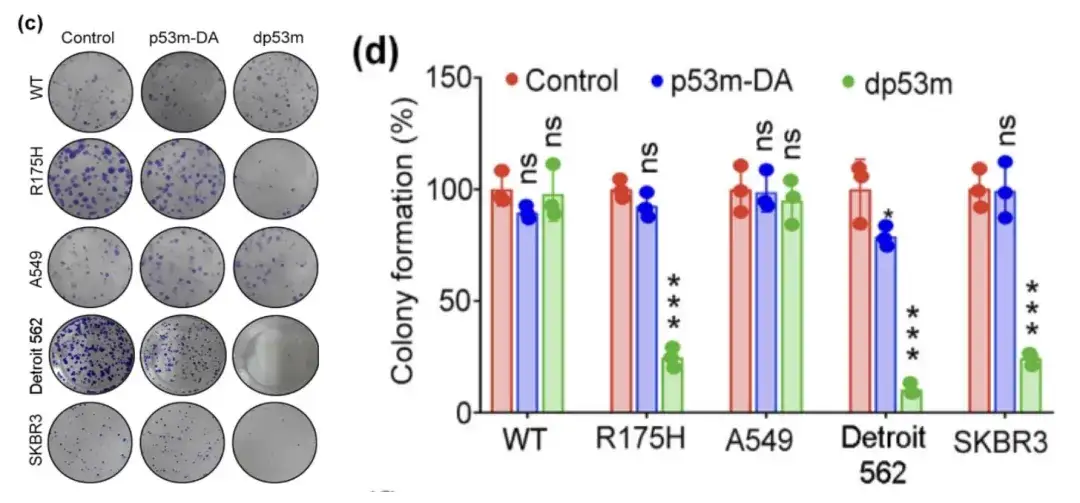
* dp53m inhibits colony formation in H1299-p53-R175H, Detroit 562, and SKBR3 cells
* No significant effect was observed in H1299-p53-WT and A549 cells
The figure above shows that dp53m exhibits a significant proliferation inhibitory effect in cancer cells expressing p53-R175H (H1299-p53-R175H, Detroit 562, and SKBR3), while cells expressing p53-WT are basically unaffected.This suggests that dp53m has great therapeutic potential in cancer cells carrying the p53-R175H mutation.
Experiment 2: dp53m inhibits the growth of p53-R175H-induced tumors in vivo
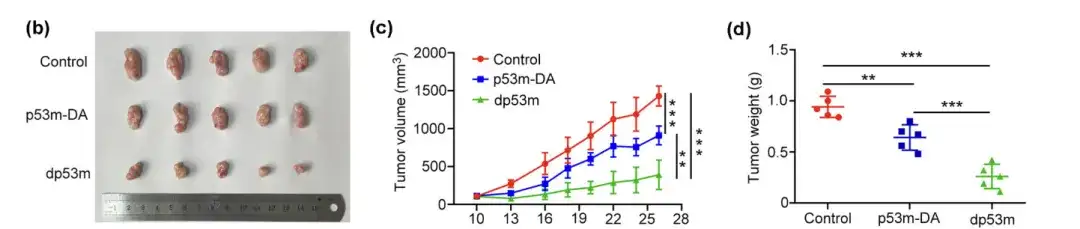
(b) Macroscopic appearance of isolated tumors after treatment
(c) Tumor volume measured at the indicated time
(d) Determination of final tumor weight
The researchers randomly assigned tumor-bearing BALB/c mice to three groups and administered p53m-DA, dp53m, or saline as a control intravenously. Compared with the control group or p53m-DA,dp53m significantly inhibited tumor growth.
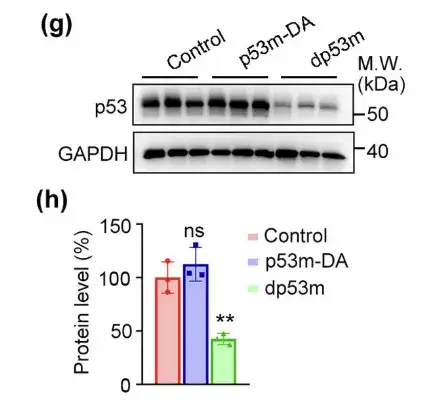
As expected,dp53m effectively reduced the expression level of p53-R175H in tumors.
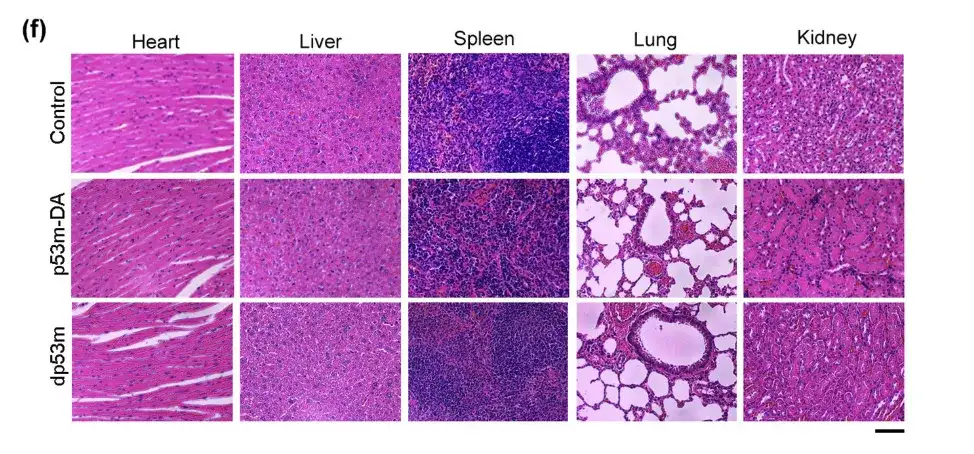
In the upper figure, there were no significant differences in the histology of heart, liver, lung, spleen, and kidney tissues among the three groups of mice.This suggests that there is no toxicity from dp53m in vivo.
In summary,dp53m can inhibit the growth of p53-R175H-induced tumors without obvious toxicity.
In addition, considering that the p53-R175H mutation is associated with resistance to chemotherapy drugs such as cisplatin, the researchers also analyzed the effect of dp53m on the anti-tumor activity of cisplatin. The results showed that there is a strong synergistic interaction between dp53m and cisplatin.That is, dp53m can enhance the sensitivity of p53-R175H cancer cells to cisplatin.This provides an "invincible partner" for cancer treatment.
Strengthening the anti-cancer defense line, scientists build a road of hope together
Cancer is a disease caused by uncontrolled cell growth, and its incidence has increased significantly with the global aging population, increased environmental pollution, and changes in lifestyle habits. According to data from the International Agency for Research on Cancer (IARC), the number of new cancer cases worldwide is estimated to increase from approximately 19.29 million in 2020 to approximately 30.23 million in 2040, an increase of 56.71%.
In the global battle against cancer, researchers play a key role. The three corresponding authors of this study are:Mr. Wu Sijin from Huihu School of Pharmacy of Xi'an Jiaotong-Liverpool University, Professor Xie Songbo and Professor Zhong Diansheng from Tianjin Medical University General Hospital are outstanding representatives in this field.
Among them, Professor Wu Sijin focuses on the research and development of new targeted drugs. His current research focuses on the use of computer-aided drug design and discovery, using simulation methods such as modeling, pharmacophore analysis, docking, and molecular dynamics (MD) to identify new therapeutic drugs. In his 2022 study, he found that upregulated SOST expression was associated with poor prognosis in breast cancer patients, and SOST activated downstream signaling pathways to promote breast cancer cell proliferation and bone metastasis. Through computational screening, the study identified a candidate therapeutic compound S6 that can disrupt the interaction between SOST and STAT3 to inhibit STAT3 phosphorylation and reduce breast cancer bone metastasis.
Paper address:
https://pubmed.ncbi.nlm.nih.gov/36581888
In addition, Professor Xie Songbo's main research direction is targeted protein degradation and drug delivery. In his 2023 study, he proposed that by using aptamers as "targeted warheads", a new strategy can be used to induce the degradation of "undruggable" proteins. To confirm this concept, the researchers selected oncogenic nucleolin (NCL) as the target, and eventually generated a series of NCL degraders. dNCL#T1 induced NCL degradation in a way that depended on the ubiquitin protease system, which could inhibit NCL-induced breast cancer cell proliferation. This study not only provides a new perspective for protein degradation, but also lays a solid foundation for the development of therapeutic drugs for diseases such as cancer.
Paper address:
https://pubmed.ncbi.nlm.nih.gov/36608275
Finally, Professor Zhong Diansheng has also conducted in-depth research in the fields of early diagnosis, chemotherapy, targeted therapy and anti-angiogenic therapy of lung cancer, and has published more than 90 academic papers. In his 2024 study, he found that CBX4 promoted the proliferation of lung adenocarcinoma (LUAD) by upregulating PHGDH expression and serine biosynthesis, and inhibited LUAD metastasis by inhibiting ZEB2 transcription. This discovery helps to understand the interaction between CBX4 and epigenetic factors, and provides inspiration for potential treatment approaches for LUAD.
Paper address:
https://www.nature.com/articles/s41419-024-06745-z
In addition to Teacher Wu Sijin, Professor Xie Songbo and Professor Zhong Diansheng, there are many scientists and doctors who are silently dedicated and working hard. We look forward to the day when cancer will no longer be a nightmare for mankind as science and technology continues to advance.